
Polynucleotides and PDRN are DNA-derived biopolymers gaining recognition for their potential in dermatology. Both are classified as deoxyribonucleotide-based polymers sourced mainly from salmon gonads and have been used in clinical settings. The global polynucleotide market has been emerging and expanding faster. These DNA-based treatments work through specific molecular mechanisms that include adenosine receptor activation and tissue repair support. We will get into the science behind their therapeutic effects and compare their molecular structures. This exploration of evidence-based applications in regenerative dermatology will help you make informed clinical decisions.
The Science Behind DNA-Based Skin Treatments

Image Source: Elixir Skin Training
What Are Polynucleotides and PDRN
Both polynucleotides and PDRN are nucleic acid-based biopolymers composed of nucleotide monomers connected via phosphodiester bonds [1]. The difference lies in their polymer chain length and molecular weight parameters. PN refers to longer DNA fragments extracted from the testes of fish such as salmon or trout, while PDRN consists of shorter chains extracted from fish sperm [2].
Both feature a comparable deoxyribonucleic acid backbone. PDRN shows lower viscoelasticity than PN due to its reduced molecular weight, whereas PN forms a more porous, viscous, three-dimensional structure [2]. This compositional difference affects their biological behavior and mechanisms of action in clinical applications.
Nucleotide Structure: Building Blocks Explained
Nucleotides are organic molecules containing monomeric units of nitrogen and phosphate bases combined with a sugar molecule [3]. The sugar component in DNA is deoxyribose, a pentose sugar that distinguishes it from RNA [3]. Nitrogenous bases include purines (adenine and guanine) and pyrimidines (thymine and cytosine), with the pairing of one purine with a pyrimidine base forming the genetic code [3].
These nucleotides serve as the building blocks of genetic material. Each nucleotide is separated by merely 0.34 nanometers. Yet DNA contained within a single human cell reaches two meters in length when extended [2]. This immense volume of information is folded into a microscopic nucleus, representing the architectural complexity that PDRN and PN employ for therapeutic purposes.
Molecular Weight Parameters: 50-1500 kDa Range
The molecular weight of polynucleotides and PDRN ranges between 50 and 1500 kilo Daltons [2][3][2]. Recent studies have expanded this range to cover fragments as small as 1 kDa and as large as 10,000 kDa [4]. The ‘Marques Polynucleotide Cutoff’ was proposed to distinguish PN having molecular weights of 1500 kDa or greater from PDRN with molecular weights below 1500 kDa [3][4]. This addresses the ambiguity in terminology.
PDRN extracted from salmon contains shorter, linear, single-stranded chains with a double helix spatial structure. The length ranges from 50 to 2000 base pairs [3]. The classic PDRN molecular weight of 132 kDa is formed through regulated purification and sterilization processes [5]. These molecular weight variations affect clinical outcomes and therapeutic applications by a lot.
Salmon-Derived DNA: Compatibility with Human Tissue
Salmon-derived PDRN is sourced from sperm harvested from chum salmon (Oncorhynchus keta) and rainbow trout (Oncorhynchus mykiss) [6][5][2]. The testicles of adult male salmon are filled with sperm, and the DNA content reaches 7.5% of its wet weight [3]. Salmon DNA exhibits over 90% similarity in protein-coding sequences with human DNA, a level of homology that stands in clear contrast to plant-derived DNA [2].
This biocompatibility stems from salmon being a vertebrate-derived biomaterial with striking genetic proximity to humans [2]. Salmon spermatozoa as a source is unique due to its immune-privileged status. This is attributed to high purity without the additives of proteins or lipids [3]. The purification process can achieve up to 90% DNA purity through high-temperature sterilization at 121°C [6]. PDRN is thermoresistant and allows classical moist heat treatment procedures in an autoclave to ensure maximum microbiological safety [6].
The extraction process targets male reproductive tissue during spawning phase. Energy is redirected toward reproduction at this time, leading to rapid expansion of gonadal tissues densely packed with sperm cells containing condensed DNA [2].
Molecular Mechanisms at the Cellular Level
A2A Receptor Binding and Signal Transduction
PDRN exerts its biological effects through two principal mechanisms: activation of the adenosine A2A receptor and provision of deoxyribonucleotide substrates through the salvage pathway [2]. The pharmacological properties of PDRN are mediated through adenosine receptor binding that targets the A2A subtype among the four known adenosine receptor variants (A1, A2A, A2B, and A3) [3]. This selective activation occurs with DNA-derived molecules within the molecular weight range of 50 to 1500 kDa [3].
Adenosine receptors function as G protein-coupled receptors that the nucleoside adenosine activates [3]. The A2A receptor is classified as a high-affinity receptor and responds to low nanomolar concentrations of adenosine [3]. Endogenous nucleases break down PDRN, and the resulting adenosine molecules bind to A2A receptors on fibroblasts, endothelial cells and immune cells [2]. Receptor engagement stimulates Gs protein and releases cyclic adenosine monophosphate (cAMP) [3]. This signal transduction suppresses both nuclear factor kappa beta (NF-κB) and mitogen-activated protein kinase (MAPK) pathways while it stimulates transforming growth factor-beta signaling cascades [3].
Salvage Pathway for DNA Synthesis Support
The nucleotide salvage pathway enables efficient reutilization of bases and nucleosides that DNA and RNA catabolism releases. This contrasts sharply with energy-intensive de novo synthesis [7]. Then, only a small fraction of nucleotides turned over daily are degraded and lost to the body [8]. This pathway proves especially critical when you have injured or hypoxic tissues where de novo synthesis is compromised and salvage becomes the predominant source of nucleotide precursors for DNA synthesis [7].
PDRN-derived oligonucleotide fragments undergo sequential hydrolysis into nucleosides and nucleotides. Cells then recycle these into the DNA of reparative cells [7]. Cells in damaged or ischemic tissues achieve DNA repair and replication more easily by utilizing this energy-efficient salvage route instead of ATP-intensive de novo pathways, therefore accelerating tissue repair [2]. This incorporation restores proliferative capacity and supports ongoing tissue regeneration [7].
Gene Expression Modulation Effects
A2A receptor activation by PDRN modulates multiple signaling cascades. It suppresses production of pro-inflammatory cytokines including tumor necrosis factor-alpha and interleukin-6 while increasing anti-inflammatory signals [2]. The pathway downregulates matrix metalloproteinase-1 (MMP-1) and initiates collagen synthesis cascades that enhance collagen production in dermal fibroblasts [7]. A2A activation upregulates vascular endothelial growth factor (VEGF) and angiopoietin-1, both facilitating neovascularization in healing tissues [2].
PDRN stimulates expression of critical bone-forming genes including RUNX2 and osteocalcin in osteogenic contexts [2]. Collagen type I alpha 1 (COL1A1) serves as an early marker of osteoblast differentiation and represents one of the first genes expressed when progenitor cells commit to osteogenic lineage [2].
Cellular Proliferation Triggers
PDRN stimulates proliferation of skin fibroblasts through involvement of A2 purinergic receptor subtypes [3]. The binding of adenosine to A2A receptors initiates cascades that result in collagen synthesis [2]. A dose-dependent increase in collagen synthesis occurs with polynucleotide application, an effect absent in cells treated with hyaluronic acid [2]. More, PDRN promotes growth of cultured human osteoblasts and increases alkaline phosphatase activity, a marker of osteoblastic function [2].
Comparing PN and PDRN: Therapeutic Differences
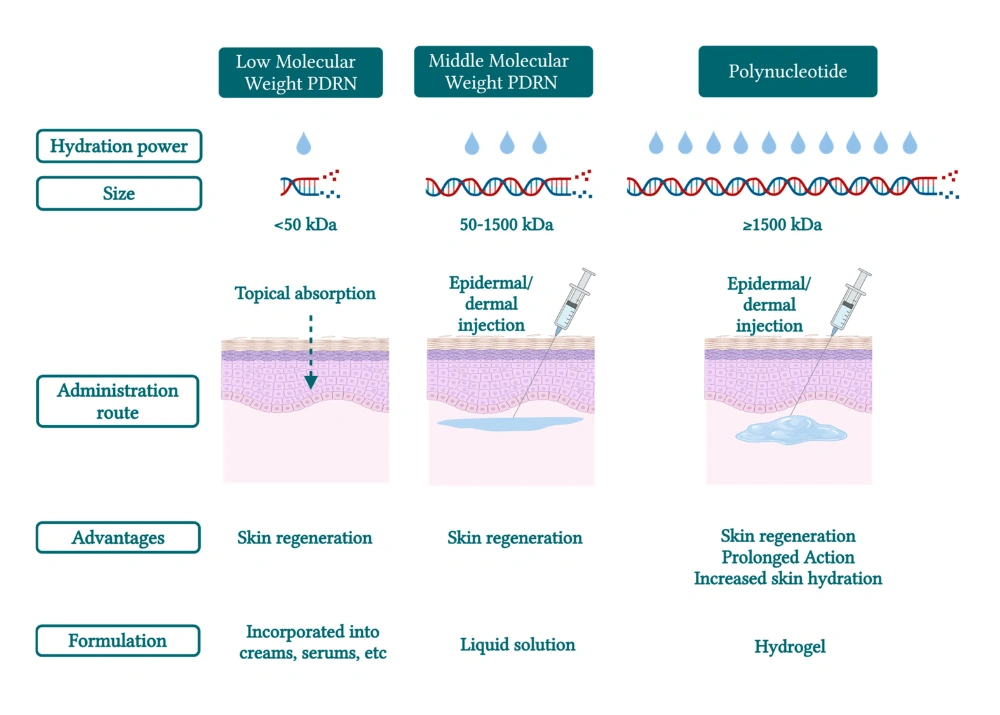
Image Source: The Esthetic Studio
Chain Length Impact on Clinical Outcomes
The therapeutic difference between polynucleotides and PDRN begins with molecular architecture. PN features longer nucleotide chains with molecular weights at or exceeding 1500 kDa, whereas PDRN consists of shorter chains that contain 10 to 30 nucleotides with molecular weights below 1500 kDa [3]. This cutoff, known as the Marques Polynucleotide Cutoff, establishes clear differentiation in product properties and clinical behavior [3].
Longer PN fragments provide more surface area and binding sites for fibroblast interaction. This enhances cellular stimulation and attachment. PDRN’s shorter chains, however, penetrate skin efficiently and mimic human DNA structure more closely. This ensures better compatibility with skin cells [9]. PN’s extended chain length translates to more robust collagen stimulation by increasing fibroblast metabolic activity, while PDRN promotes collagen formation in damaged or compromised skin [9].
Viscoelastic Properties and Hydration Capacity
PN exhibits higher viscosity and forms a distinct framework structure absent in PDRN formulations [3]. PN contains high water content and creates a hydrogel that improves hydration and restores skin viscosity [2]. These viscoelastic and hydrophilic properties position PN as a potential alternative to hyaluronic acid in various applications [2].
Degradation kinetics differ between formulations. A liquid PDRN preparation degrades faster than a PN-based hydrogel, as nucleases access a much smaller surface area in the latter [6]. PN-based hydrogels then present slower degradation kinetics while ensuring prolonged skin stimulation for sustained results [6].
PN for Structural Support vs PDRN for Active Regeneration
PN functions as a structural and rheological support agent. It provides long-lasting effects through sustained regeneration [3][2]. Its higher-order polymer structure contributes to potent anti-inflammatory action and helps modulate immune responses and reduce skin inflammation [9]. This makes PN especially effective for detailed anti-aging treatments and full-face skin regeneration [9].
PDRN, on the other hand, is characterized as a pharmacologically active molecule with specific mechanisms of action [2]. It acts on regeneration and repairing damaged DNA, increasing fibroblast activity, inducing angiogenesis, and providing anti-inflammatory effects [3]. PDRN excels in targeted skin repair, making it ideal for addressing acne scars, fine lines, stretch marks, and post-procedure recovery [9].
Duration of Effects and Dosing Protocols
Clinical studies demonstrate specific PDRN dosing protocols. Diabetic patients received 5.625 mg diluted in 3 ml via intramuscular injection five days per week and perilesional injection two days per week for eight weeks [7]. This regimen nearly doubled the rate of complete healing of diabetic foot ulcers compared to placebo [7]. The drug’s half-life is three hours and remains unaffected by dosage [7].
Dermatological Applications and Treatment Protocols
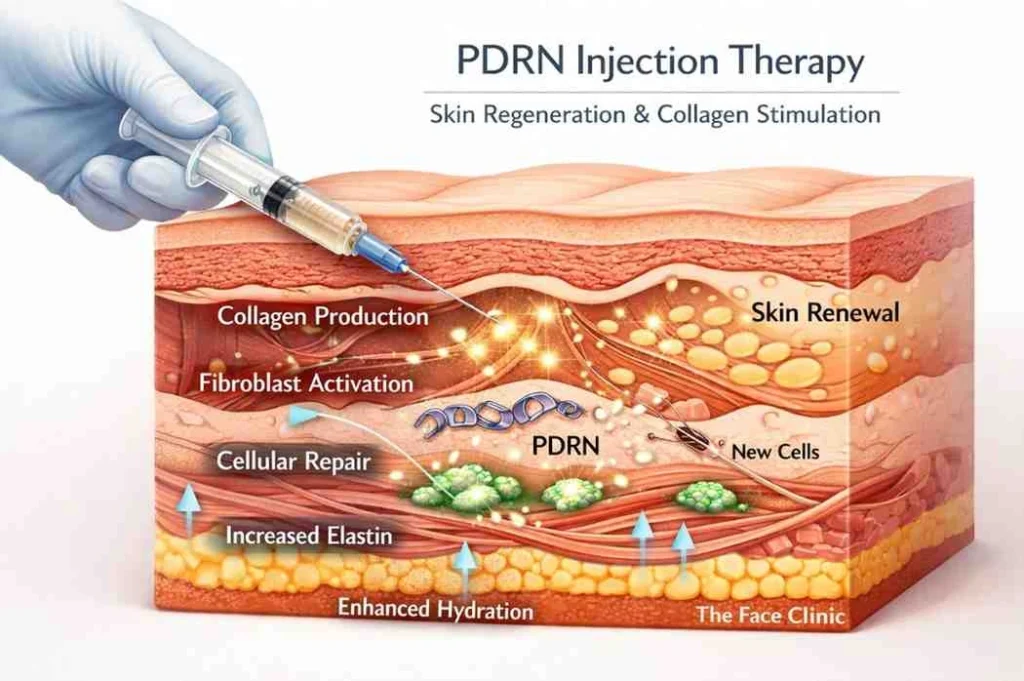
Image Source: Dr Reema Arora
Evidence-Based Indications in Skin Health
Clinical applications of polynucleotides and PDRN extend across multiple dermatological conditions. Korean cosmetic physicians have used PN to treat facial erythema resulting from inflammatory facial dermatosis, with 84.4% reporting its use for this indication [8]. Among those practitioners, 88.1% found the treatment highly effective or effective [8]. About 66.4% used PN to treat erythema caused by repeated laser or microneedling radiofrequency procedures [8].
The main indications center on skin quality improvement rather than volumization. Fine lines linked to reduced skin quality, early laxity, texture concerns and delicate periocular skin represent ideal treatment targets. PDRN showed a 61.4% reduction in periorbital wrinkles after 12 weeks in a study with 218 Asian subjects [1]. PDRN achieved complete wound closure in 37.3% of patients with diabetic foot ulcers compared to 18.9% in the placebo group after eight weeks [1].
Combination with HA and Other Injectables
Combination protocols improve therapeutic outcomes through cooperative mechanisms. About 55.4% of clinicians used PN in combination with hyaluronic acid to treat facial erythema [8]. The pairing proves especially effective because HA provides immediate hydration while PDRN initiates regenerative processes. Sequential injection approaches involve administering PDRN first followed by HA skin booster in the same session, or alternating treatments every two weeks [10].
About 28.1% combined PN with botulinum toxin to treat post-laser erythema [8]. Practitioners also integrated PN with lesion targeting devices (36.2%) or regenerative devices (31.6%) [8]. Crosslinked HA-PDRN composite fillers demonstrate improved fibroblast proliferation and collagen production compared to HA alone [11].
Patient Selection Criteria
Suitable candidates include those seeking texture improvement, natural progressive results, or treatment of delicate areas unsuitable for volumizing fillers. Contraindications include pregnancy, breastfeeding, active infection and certain inflammatory skin conditions. Fish allergy requires careful thought given the salmon-derived source material.
Expected Outcomes and Timeline
Results show around one month after the final treatment, with continued improvement over subsequent months. Peak effects occur approximately three months post-treatment as collagen regeneration progresses. Immediate hydration effects appear within days, while structural improvements develop over time.
Treatment Frequency Recommendations
The most common clinical protocol involves injecting 2cc of PN every three to four weeks for a total of three to four sessions [8]. Treatments are spaced two to four weeks apart during the initial cycle. Maintenance sessions occur every six to twelve months to sustain results and prevent regression in collagen production.
Critical Considerations for Clinical Practice

Image Source: Isaac Luxe
Addressing Mechanism Uncertainty
Despite clinical adoption, the exact mechanism through which PDRN regulates certain effects remains incompletely understood and requires future investigation [12]. The distinct characteristics of PDRN and PN are underrecognized. This results in clinical misapplications and suboptimal treatment outcomes [2]. This knowledge gap underscores the need for rigorous, mechanistically driven research to clarify the distinct pharmacodynamic and biological properties of each compound [2].
Quality Control and Product Variability
Challenges arise due to inadequate operating protocols and structured guidelines for sourcing and manufacturing [3]. Standardization of doses, mode of delivery, exact downstream effects, safety profile, drug interactions, sourcing, extraction, and storage all need elucidation to accept these as therapeutic agents with established quality and reliability [3]. Pharmaceutical companies have established proprietary platforms for extraction and purification. They use these technologies to brand their products [2].
Energy Cost of Catabolism
PDRN undergoes degradation by unspecific plasma DNA nucleases or by nucleases bound to cell membranes. This leads to oligo and mononucleotides [7]. PDRN fragments are then excreted in urine (about 65%) and to a lesser extent in feces [7].
Fish Allergy and Safety Concerns
Fish represents one of eight major allergenic foods [13]. But not all patients with fish allergy develop reactions to PN or PDRN [3]. Products are purified by filtration during extraction [3]. Prior patch testing with the drug may help identify susceptible patients [3]. PDRN is extracted and purified at high temperatures. This recovers more than 95% pure active substances with inactivated proteins and peptides, guaranteeing safety due to absence of immunological effects [12].
Future Research Directions
More controlled trials and extrapolation of in vitro findings to translational medicine need undertaking [3]. A systematic review concluded that though the potential of PNs is most important, current evidence remains fraught with inconsistencies [3].
Conclusion
Polynucleotides and PDRN represent a promising frontier in regenerative dermatology. They are backed by specific molecular mechanisms that include A2A receptor activation and DNA salvage pathways. We covered the fundamental structural differences between these compounds, with PN offering sustained structural support while PDRN drives active tissue repair. Their salmon-derived origin ensures biocompatibility with human tissue.
Clinical adoption continues to grow, but note that standardization challenges and mechanistic uncertainties persist. Future research must establish quality control protocols and dosing guidelines. These DNA-based treatments hold most important potential for advancing esthetic and therapeutic dermatology practices as evidence continues to evolve.
Stay at the forefront of dermatological innovation. Contact our technical team at CL Cosmetic Industries via the WhatsApp/live chat button on the homepage or fill-up the form on the homepage for more scientific insights on Polynucleotides and PDRN applications & ways to get started with your skincare range now.
Key Takeaways
Understanding the molecular science behind polynucleotides and PDRN empowers dermatologists to make informed treatment decisions and optimize patient outcomes in regenerative esthetics.
• Molecular weight determines function: PN (>1500 kDa) provides structural support and hydration, while PDRN (<1500 kDa) drives active cellular regeneration through A2A receptor activation.
• Salmon-derived DNA offers superior biocompatibility: Over 90% genetic similarity to human DNA ensures excellent tissue compatibility, with purification processes achieving 95% purity and minimal allergenic risk.
• Evidence supports specific clinical protocols: Optimal results require 3-4 sessions spaced 2-4 weeks apart, with effects peaking at 3 months as collagen regeneration progresses.
• Combination therapies enhance outcomes: Pairing with hyaluronic acid or other treatments creates synergistic effects, with 55% of practitioners successfully combining PN with HA for improved results.
• Quality control remains critical: Standardization challenges and mechanistic uncertainties require careful product selection and adherence to established protocols for consistent therapeutic outcomes.
The therapeutic potential of DNA-based treatments continues expanding, but success depends on understanding their distinct mechanisms and implementing evidence-based protocols tailored to individual patient needs.
FAQs
Q1. What is the main difference between polynucleotides (PN) and PDRN in dermatology treatments? The primary difference lies in their molecular weight and chain length. Polynucleotides have molecular weights of 1500 kDa or greater with longer DNA chains, providing structural support and sustained hydration. PDRN has molecular weights below 1500 kDa with shorter chains (10-30 nucleotides), functioning as a pharmacologically active molecule that drives cellular regeneration and tissue repair through A2A receptor activation.
Q2. How do PDRN treatments work at the cellular level to improve skin? PDRN works through two main mechanisms: it activates adenosine A2A receptors on fibroblasts and endothelial cells, triggering collagen synthesis and reducing inflammation, and it provides nucleotide building blocks through the salvage pathway that support DNA repair in damaged cells. This dual action stimulates cellular proliferation, enhances collagen production, and promotes neovascularization for improved skin regeneration.
Q3. Are polynucleotide and PDRN treatments safe for people with fish allergies? While these treatments are derived from salmon DNA, the extraction and purification process occurs at high temperatures (121°C), recovering more than 95% pure active substances with inactivated proteins and peptides. This significantly reduces allergenic risk. However, patients with known fish allergies should undergo prior patch testing to identify potential sensitivity before treatment.
Q4. How long does it take to see results from polynucleotide or PDRN treatments? Results typically appear around one month after completing the initial treatment series, with continued improvement over subsequent months. Immediate hydration effects may be visible within days, but structural improvements develop gradually as collagen regeneration progresses. Peak effects generally occur approximately three months post-treatment.
Q5. What is the recommended treatment protocol for polynucleotide injections? The most common protocol involves 3-4 treatment sessions spaced 2-4 weeks apart, with each session using approximately 2cc of product. After completing the initial series, maintenance sessions are recommended every 6-12 months to sustain results and prevent regression in collagen production. This schedule allows for optimal collagen stimulation and long-lasting skin quality improvement.
References
[1] – https://www.mdpi.com/2076-3417/15/19/10437
[2] – https://pmc.ncbi.nlm.nih.gov/articles/PMC12388916/
[3] – https://jcasonline.com/polynucleotides-and-polydeoxyribonucleotides-in-dermatology-a-narrative-review/
[4] – https://pubmed.ncbi.nlm.nih.gov/39858543/
[5] – https://www.spandidos-publications.com/10.3892/mmr.2018.9539
[6] – https://www.mdpi.com/2218-273X/15/1/148
[7] – https://pmc.ncbi.nlm.nih.gov/articles/PMC5405115/
[8] – https://pmc.ncbi.nlm.nih.gov/articles/PMC10485387/
[9] – https://laboderm-skin.com/differences-between-pn-pdrn/
[10] – https://pdrn.care/combinations/pdrn-and-hyaluronic-acid/
[11] – https://www.sciencedirect.com/science/article/pii/S0014305724006566
[12] – https://www.sciencedirect.com/science/article/pii/S2096691122000723
[13] – https://pubs.acs.org/doi/10.1021/acsfoodscitech.3c00572
